6.3: Thermodynamics and kinetics - Engineering LibreTexts
SOLVED: Calculate the Tafel slopes for the anodic and cathodic reactions, and determine the Stern-Geary coefficient. Also, calculate the corrosion rate per year. F = 96,485 C mol^-1 Molecular weight of iron =
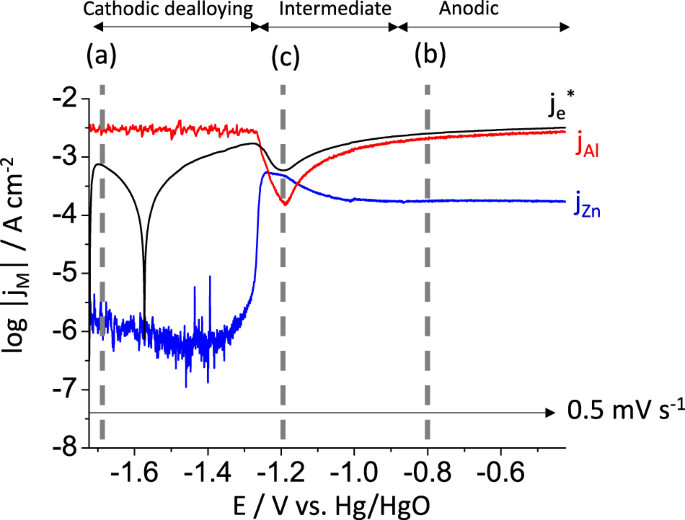
Refining anodic and cathodic dissolution mechanisms: combined AESEC-EIS applied to Al-Zn pure phase in alkaline solution
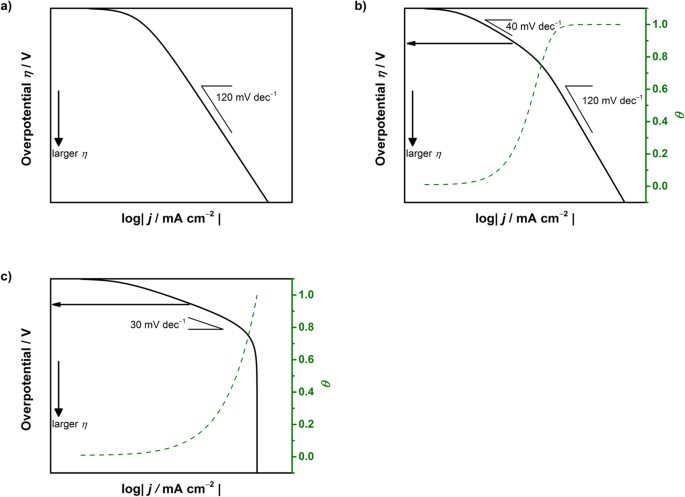
Insight on Tafel slopes from a microkinetic analysis of aqueous electrocatalysis for energy conversion
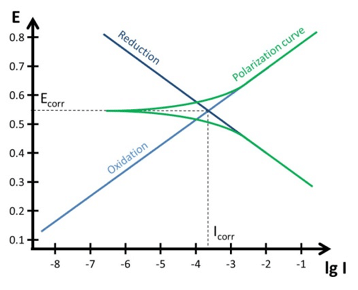
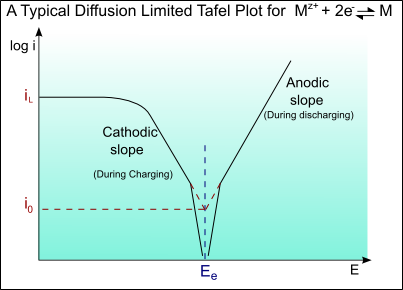
Tafel Plot and Evans Diagram - PalmSens
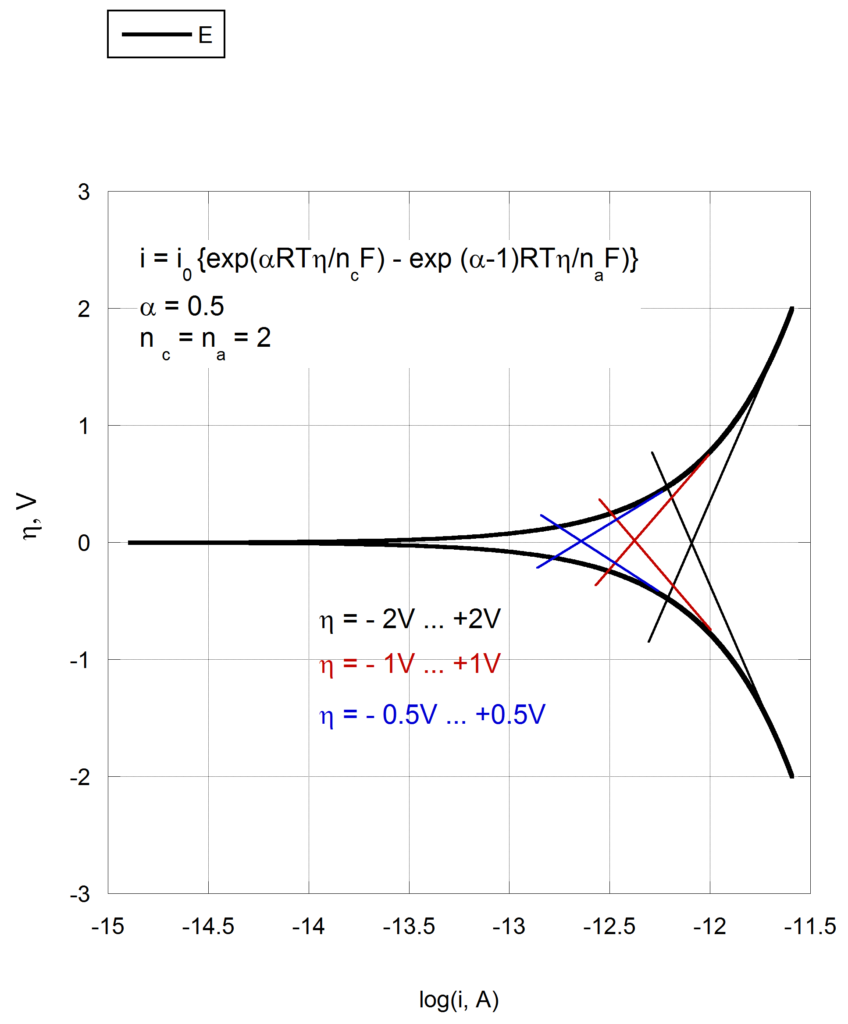
About the measurement of Tafel slopes - Personal site of Alex Lugovskoy

Solved 2. The exchange current density of the reaction 2H+ +
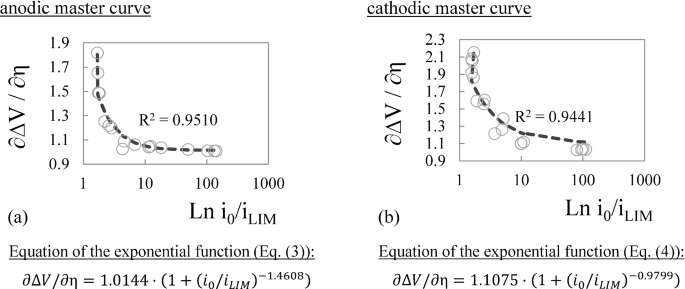
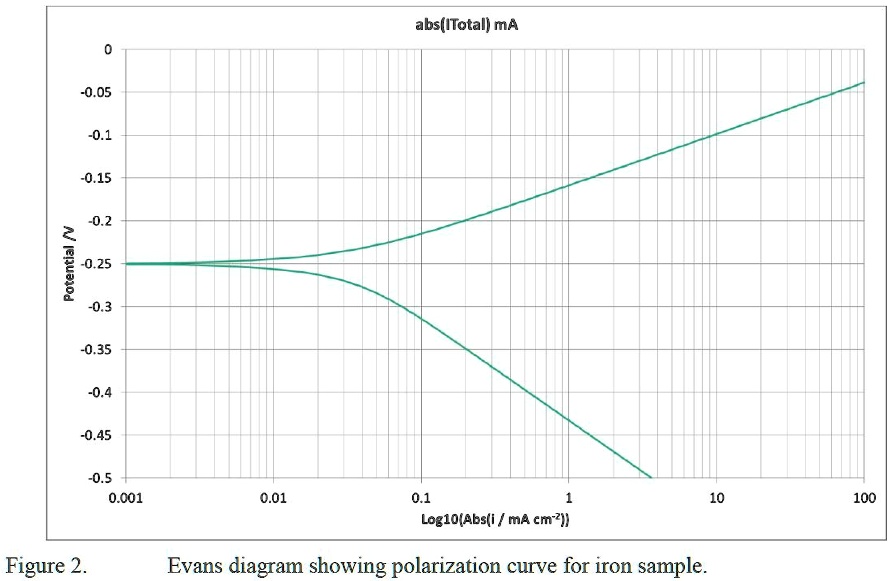
Improved Tafel-Based Potentiostatic Approach for Corrosion Rate Monitoring of Reinforcing Steel
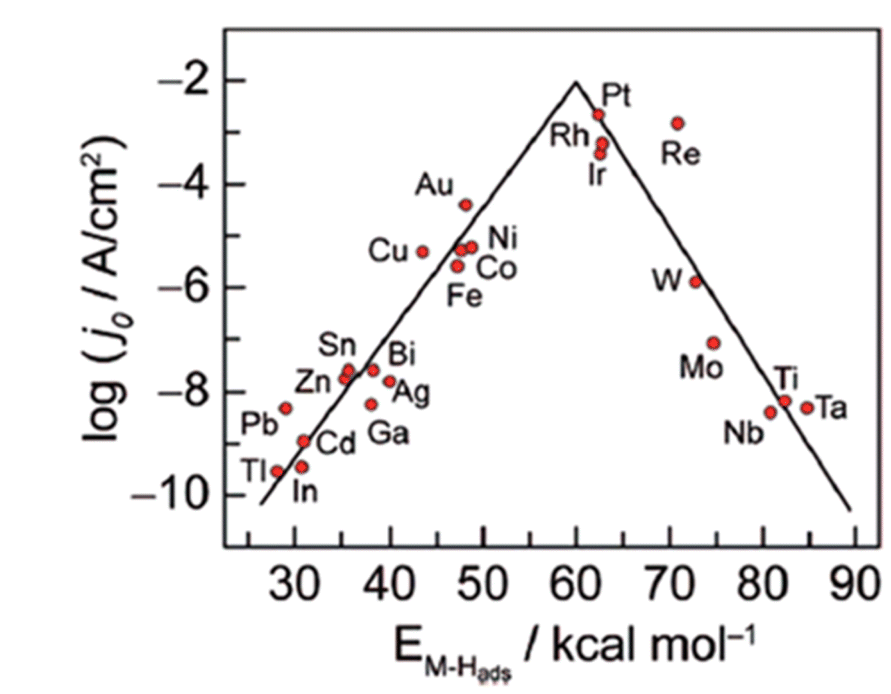
A comprehensive review on the electrochemical parameters and recent material development of electrochemical water splitting electrocatalysts - RSC Advances (RSC Publishing) DOI:10.1039/D2RA07642J
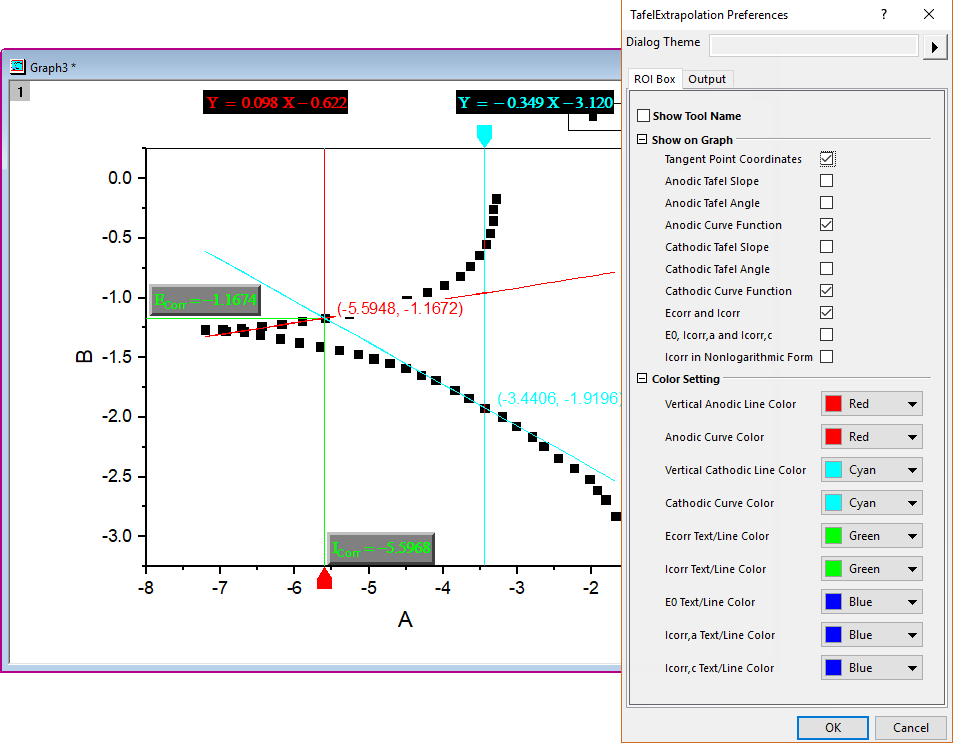
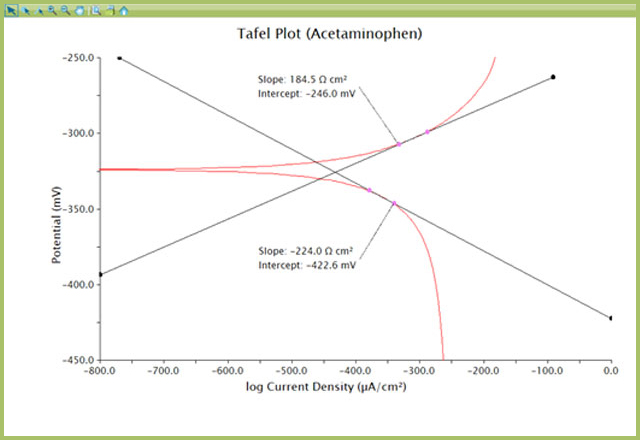
Tafel Extrapolation - File Exchange - OriginLab

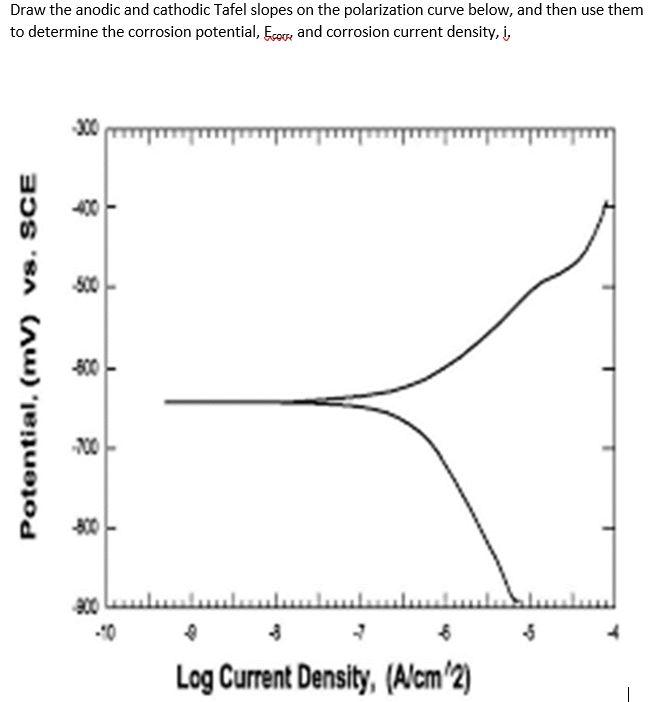
Corrosion potential E corr , anodic and cathodic Tafel slopes b a and b

SOLVED: Corrosion rate of the iron (mm/year) using the following data: Fe2+ + 2e = Fe E = -0.44 V vs SHE Anodic Tafel slope for Fe dissolution = +0.23 V/decade Fe

Anode Z N Cathode 2 H, PDF, Anode
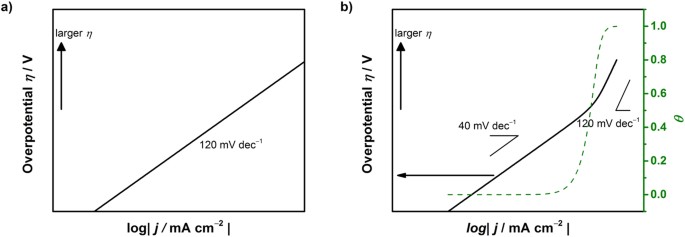
Insight on Tafel slopes from a microkinetic analysis of aqueous electrocatalysis for energy conversion

Tafel Extrapolation - an overview