The value of compression factor at the critical state of a vander waals gas is

Critical Constants and the Van Der Waals Equation of State

Gaseous State.pdf - Chemistry - Notes - Teachmint

It's a gas: critical behavior in the van der Waals gas derivation

SOLVED: State (i) the ideal gas equation (ii) the van der Waal's equation of state. Derive an expression for the compression factor; Z of a van der Waals gas in terms of
Gaseous State.pdf - Chemistry - Notes - Teachmint
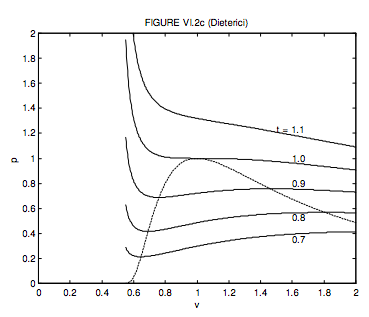
6.3: Van der Waals and Other Gases - Physics LibreTexts
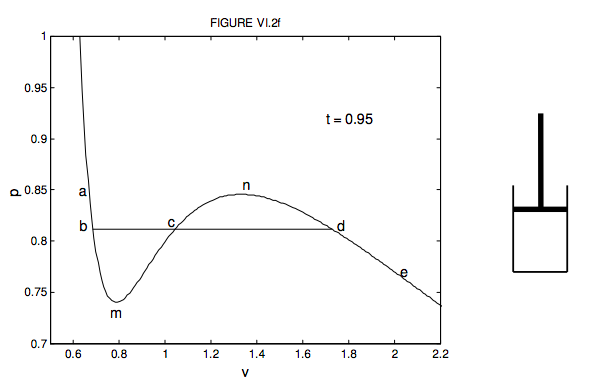
6.3: Van der Waals and Other Gases - Physics LibreTexts

1. A Choose the correct option(s) A) At low pressure (nearly 1 atm), compressibility factor H, gas is greater than 1 273 K. VB) Compressibility factor a vander Waal's gas its critical

Solved The van der Waals equation of state can be used to







