
In the following compressibility factor Z vs pressure graph at 300 K, the compressibility of CH 4 at pressure
In the following compressibility factor Z vs pressure graph at 300 K- the compressibility of CH 4 at pressure -200 bar deviates from ideal behaviourA- The molar volume of CH 4 is less than its molar volume in the ideal stateB- The molar volume of CH 4 is same as that in its ideal stateC- Intermolecular interactions between CH 4 molecules decresasesD- The molar volume of CH 4 is more than its molar volume in the ideal state

Gas Laws / Gases Behaviour, PDF, Gases

Compressibility factor - Wikipedia
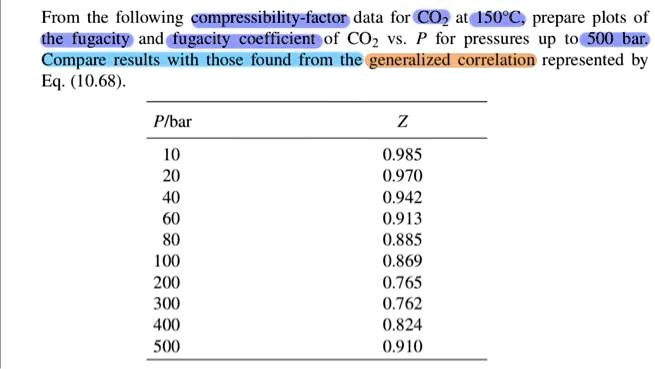
SOLVED: From the following compressibility-factor data for CO at 150°C, prepare plots of the fugacity and fugacity coefficient of CO vs. P for pressures up to 500 bar. Compare the results with

How is matter classified at micro level

Solved Molar volume of the gas calculated from ideal gas

In the following compressibility factor (z) vs. pressuer graph 300 K, the compressibility of { CH }_{ 4 } pressures <200 bar from ideal behaviur because
Assuming that the behaviour of ammonia is correctly described by the van der Waals equation near the critical point and knowing the critical molar volume of 072500 mol L1 and critical temperature of 4053 K determine the critical pressure of ammonia

KVPY-SX 2016 Chemistry Question Paper with Solutions PDF Download
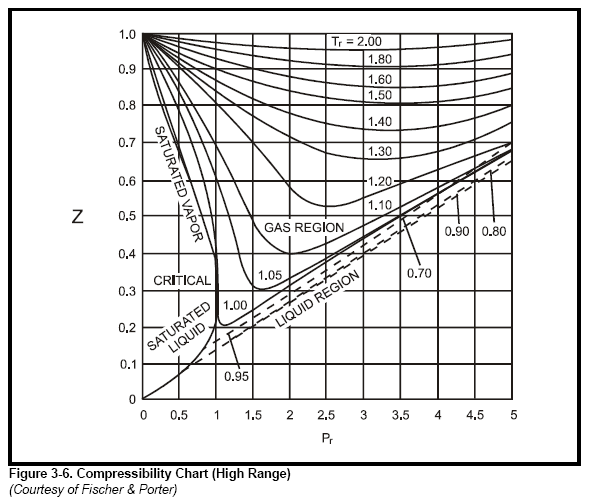
Thermo] Derivation of compressibility factor vs reduced pressure

The graph of compressibility factor (Z) vs. P for one mole of a real gas is shown in following diagram. The graph is plotted at constant temperature 2 - Sarthaks eConnect


/product/78/2100161/1.jpg?6804)


