
Share

Real Gas Effect - an overview

The Ideal Gas Law
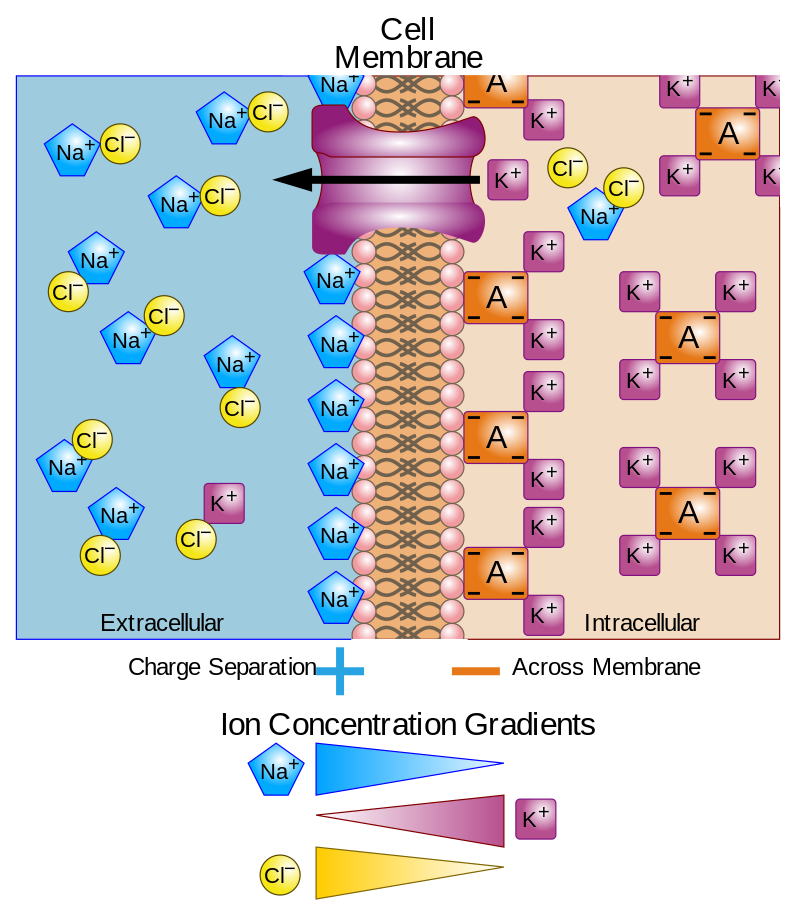
Membrane potential - Wikipedia
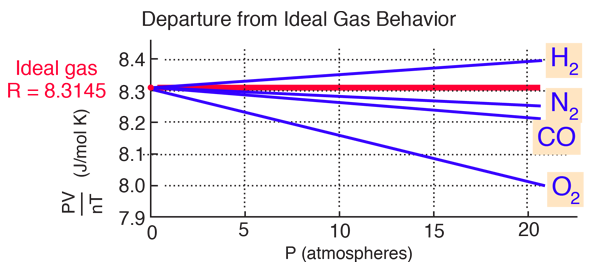
van der Waals Equation of State

Chapter 7.1: Real Gases - Chemistry LibreTexts

Performance Corrections for Compressor Maps

Deviation Of Real Gas From Ideal Gas Behavior
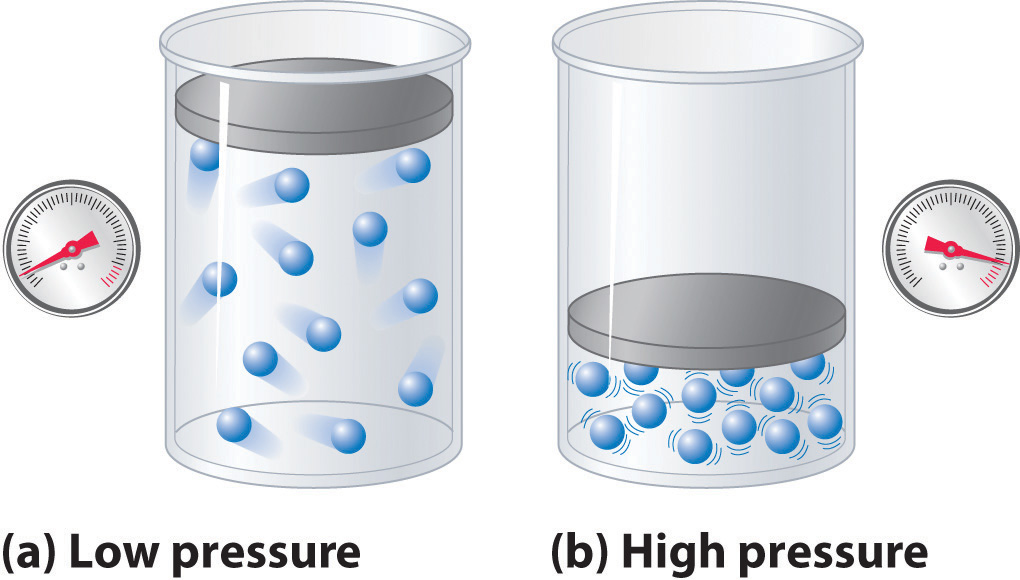
Chapter 7.1: Real Gases - Chemistry LibreTexts

Solved 4. Show that the following results are true for a

Luke Skywalker, Wookieepedia
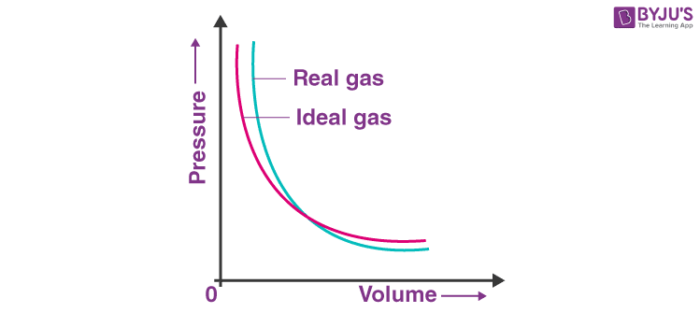
Deviation Of Real Gas From Ideal Gas Behavior
Related products
At low pressure, the van der waal's equation is written as (P+ a/V^2)V=RT . Then compressibility factor is then equal to

At a high pressure, the compressibility factor (Z) of a real gas is us

At high pressure, the compressibility factor 'Z' is equal toa)unityb) c) d)ZeroCorrect answer is option 'C'. Can you explain this answer? - EduRev NEET Question
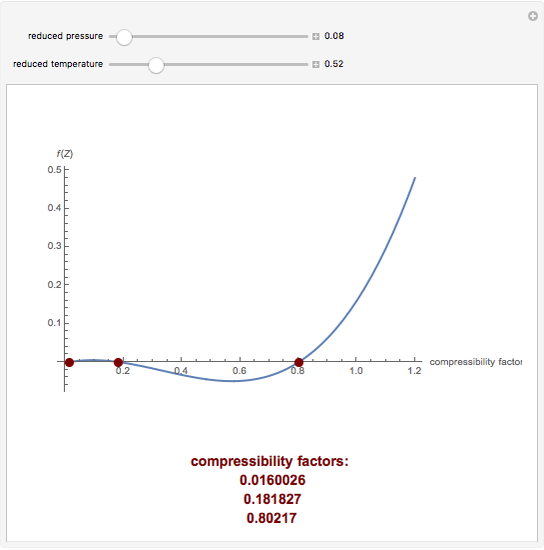
Cubic Equation of State for the Compressibility Factor - Wolfram
You may also like



