
Accelerated aging is a medical package testing method that can accurately assess packaging durability by simulating environmental factors such as heat and humidity.
The Accelerated Aging Study simulates the effects of aging, weathering and handling by subjecting sterile packaged product to conditions considered

Accelerated Aging Study

PDF) Accelerated aging system for prognostics of power semiconductor devices

Packaging Validation Process and Guidelines: Accelerated Aging
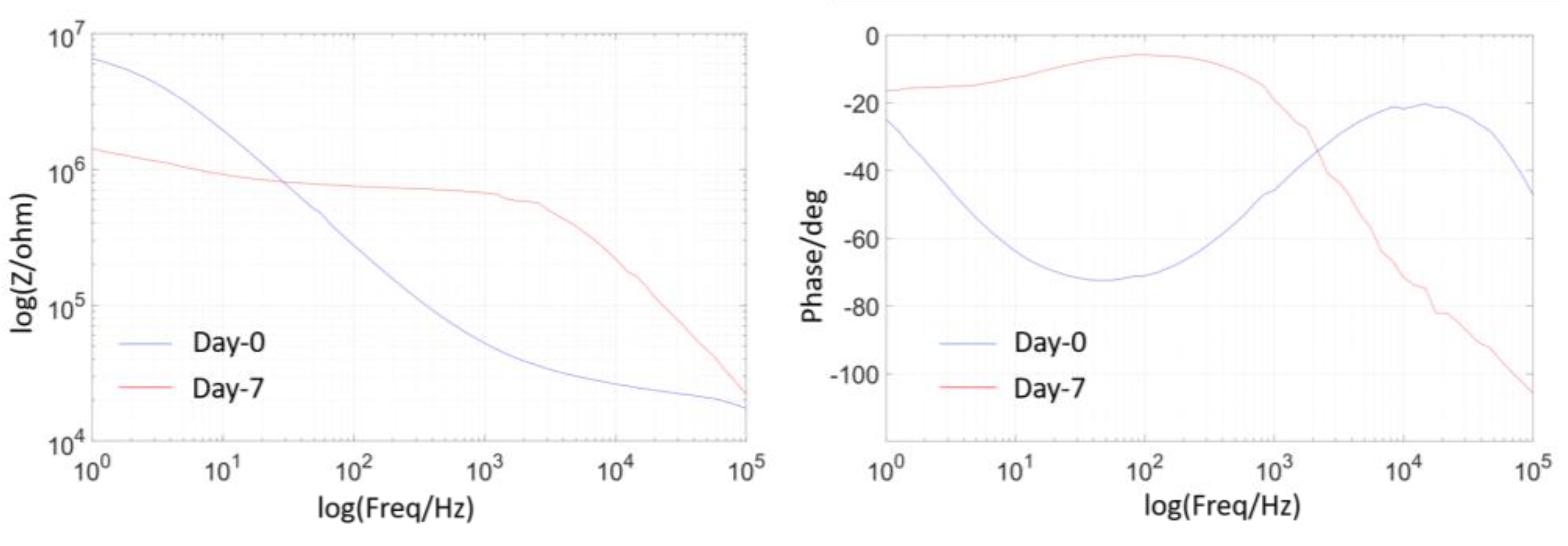
Micromachines, Free Full-Text

How to handle accelerated aging in medical device packaging 📦🕒 Learn more in our blog., Medical Device Manufacturing posted on the topic

Accelerated Aging Testing from DDL
Standard Practice for Determination of Expiration Dating for Medical Gloves

ASTM D7160-16(2023) - Standard Practice for Determination of Expiration Dating for Medical Gloves

YY∕T 0681.1-2018 Test Methods for Sterile Medical Device Packaging Part 1: Guidelines for Accelerated Aging Tests

Accelerated Aging Package Testing at Keystone Compliance







