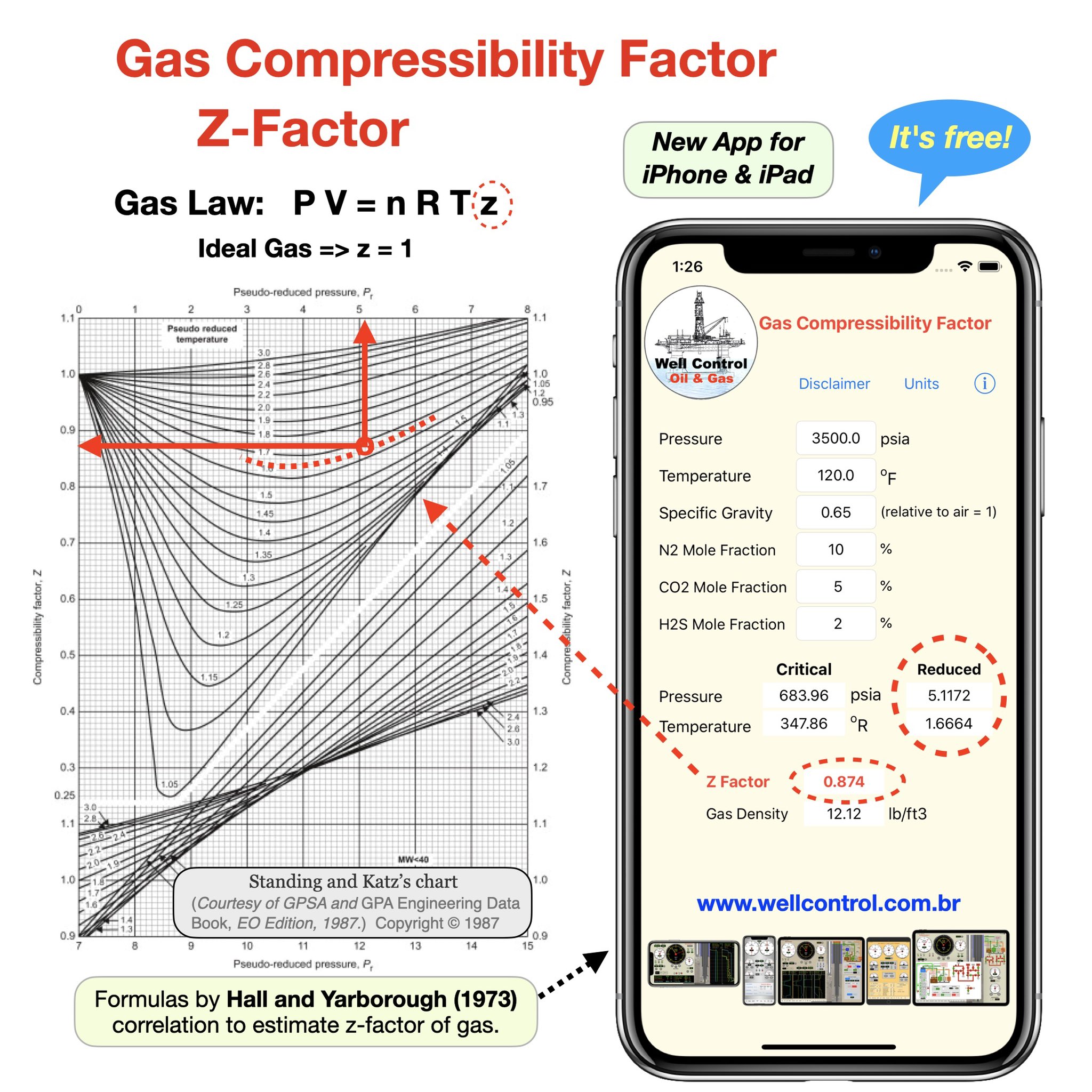
The compressibility factor Z for real gases can be rewritten

The compressibility factor for a real gas is expressed by, z =1+

SOLVED: Show that P κ=1-P((∂ln z)/(∂ P))T for a real gas where κ is the isothermal compressibility.

Real Gas Behavior The Compression Factor (Z) [Example #2]

Compressibility factor Z - Gaseous State

Compressibility Factor for Real Gas, Gaseous State, Class 11
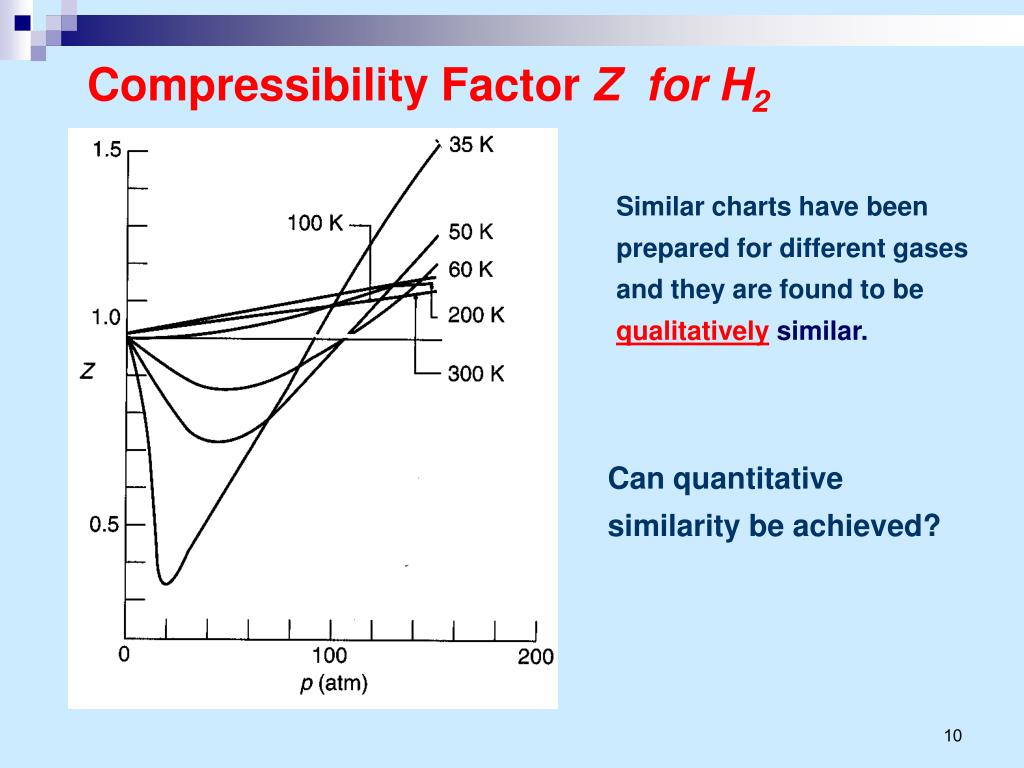
PPT - The Ideal Gas PowerPoint Presentation, free download - ID:6789672

Real Gases vs Ideal Gases & the Compressibility Factor

The given graph represents the variation of compressibility factor Z vs P for three gases A, B and C.Identify the incorrect statements.

Compressibility factor (gases) - Citizendium

Compressibility Factor of Gas Overview, Equation & Chart

Real Gas - Definition and Detailed Explanation with FAQs, Compressibility Factor for a Real Gas
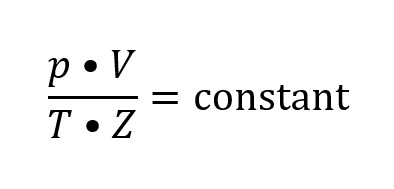
Pressure and temperature compensation in flow measurements

gas laws - Graph of compressibility factor vs pressure when real

The internal pressure (oulov),, of a real gas is related to the

Compressibility factor - Wikipedia