An ideal gas is taken from (Pi , Vi ) to (Pi , Vi ) in three different ways. Identify the process in (d) Equal work is done in Process A, B & C

SOLVED: ideal gas initially at Pi, Vi, and Ti is taken through cycle as shown below: (Let the factor n 3.7.) nf Find the net work done on the gas per cycle

In the given figure an ideal gas changes its state from `A` to state `C` by two paths `ABC` and
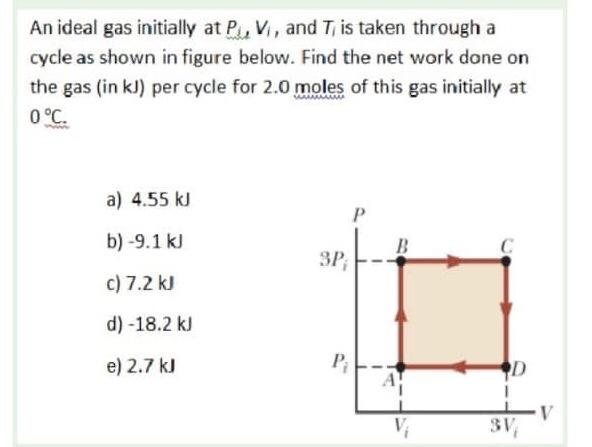
Solved An ideal gas initially at PJ, V, and Ti is taken

An ideal gas is taken around the cycle ABCA as shown in P-V diagram. The net work done by the g

An ideal gas is taken around the cycle ABCA as shown in P-V diagram. The net work done by the ga

1568 Work from PV diagram An ideal gas can have the processes shown in the PV

An ideal gas is taken from state A to state B via three different processes as shown in the pressure volume (P-V) diagram. If Q4, Q, & Q, indicates the heat absorbed

One mole of an ideal gas is taken from state A to state B by three different processes a A C B, b A D B and c A E B as

1568 Work from PV diagram An ideal gas can have the processes shown in the PV
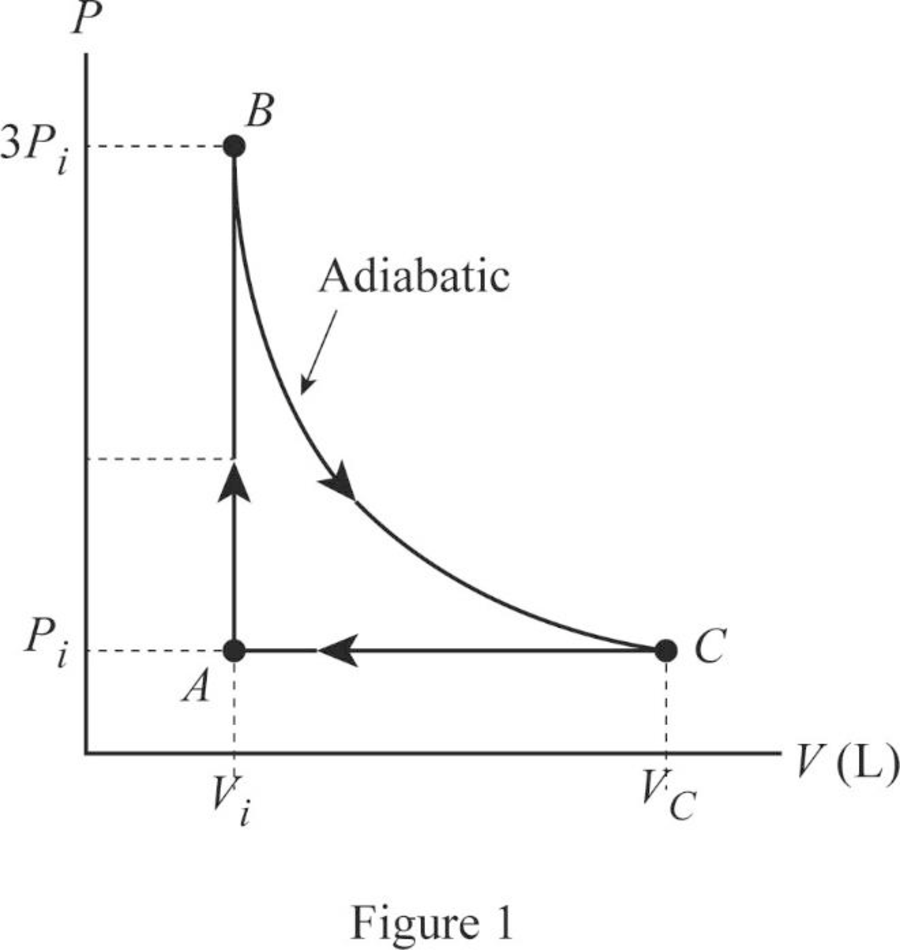
An ideal gas with specific heat ratio γ confined to a cylinder is put through a closed cycle. Initially, the gas is at P i , V i , and T i .
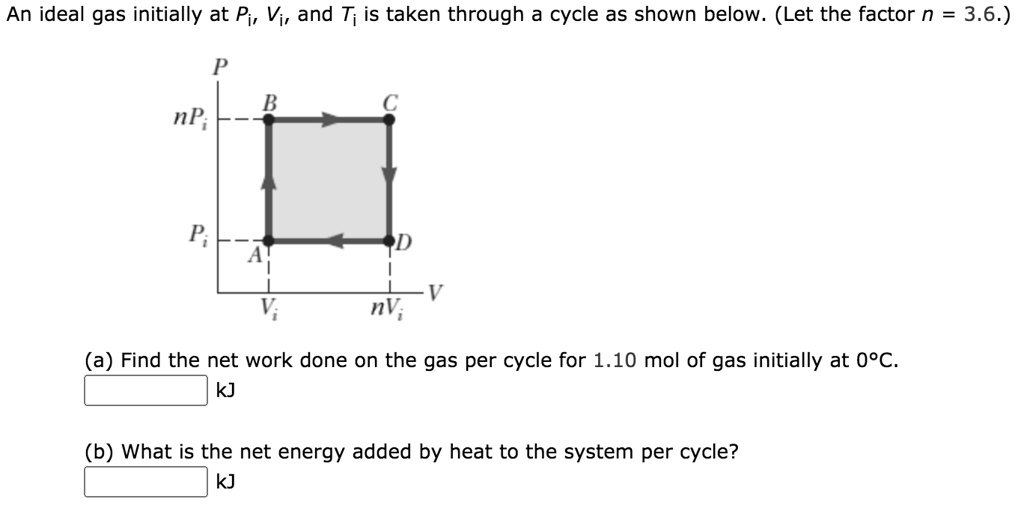
SOLVED: An ideal gas initially at Pi, Vi, and Ti is taken through a cycle as shown below. (Let the factor n 3.6.) nP; P; nV; (a Find the net work done
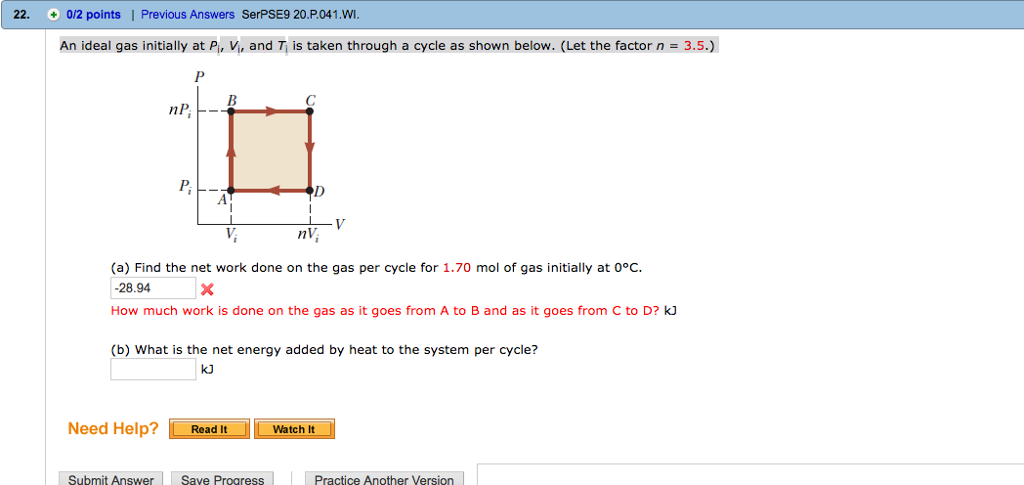
Solved An ideal gas initially at Pi, Vi, and Ti is taken
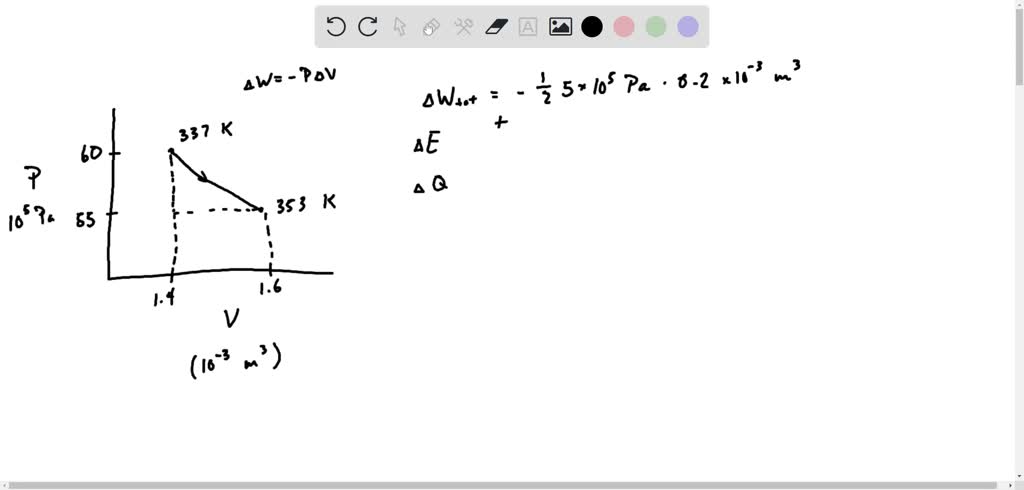
SOLVED: Using multiple models simultaneously: This FNT refers to a T = 337K involving three moles (which 60.0 process diatomic gas 2 behaves as an ideal gas). The PV curve at right

An ideal gas is taken around the cycle ABCA as shown in P-V diagram. The net work done by the ga







