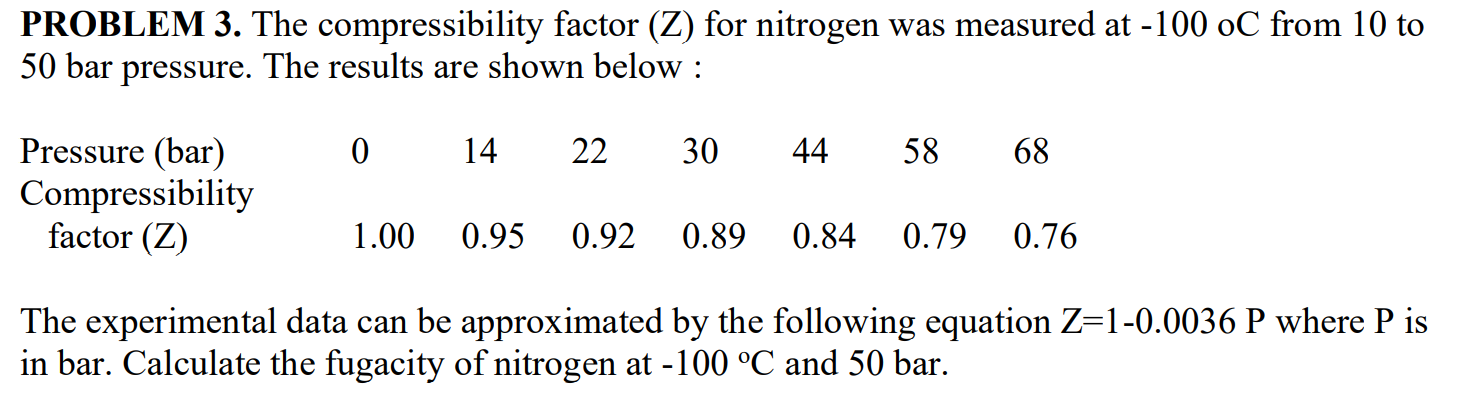

Gas Compressibility - an overview

Non-Ideal Gas Behavior Chemistry: Atoms First

SOLVED: PROBLEM 3: The compressibility factor (Z) for nitrogen was
The given graph represent the variation of z compressibility factor z=pv/nRT versis p fpr three real gases A,B,C identify only incorrect statement

Real Gas Behavior The Compression Factor (Z) [Example #2]

What is the compressibility factor (Z) for 0.02 mole of a van der Waal

Solved Problem 3 (35 marks): Calculate the compressibility

Compressibility factor for methane.

i.ytimg.com/vi/3ta9OUAC4IY/sddefault.jpg
Chapter 3 - Physical Properties of Fluids: Gas Compressibility Factor

Compressibility factor (gases) - Knowino

Compressibility factor Z = PV / nRT is plotted against pressure as shown below:What is the correct order for the liquefiability of the gases shown in the above graph? A. CO 2



.png)



